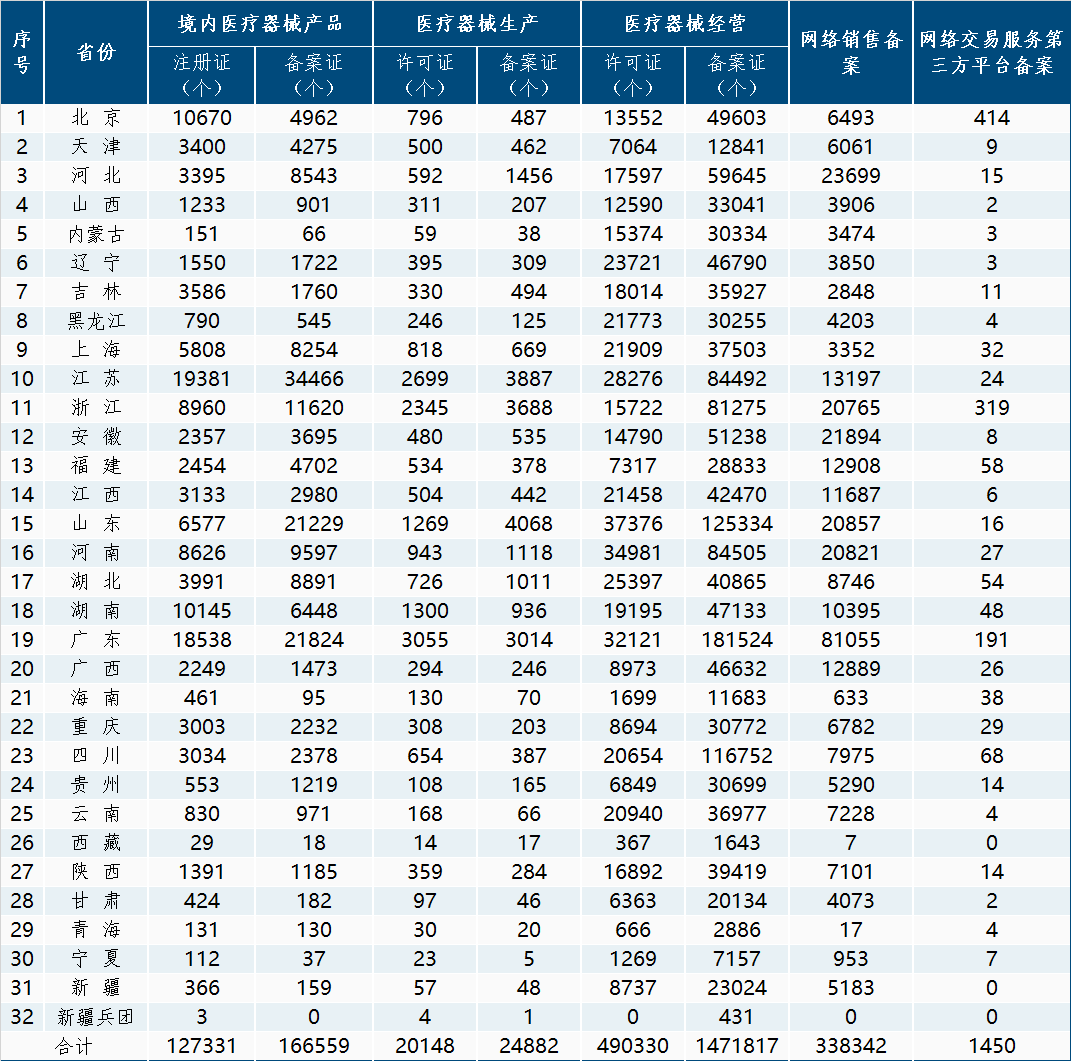
In order to further meet the needs of enterprises and the public to query medical device regulatory information, strengthen social supervision and governance, the National Medical Products Administration has compiled the registration and filing information of medical device products, medical device production enterprise licenses, medical device operation enterprise licenses, medical device online sales, and third-party platform registration information for medical device online trading services within the validity period of each province (autonomous region, municipality directly under the central government), and published it on a monthly basis.
Specific data can be queried on the medical device module of the National Medical Products Administration's data query page( https://www.nmpa.gov.cn/datasearch/home-index.html?itemId=2c9ba384759c957701759ccef50f032b#category=ylqx ).
If you have any questions about the data, please follow the error correction and maintenance methods published in the website database.
相关推荐
- 2026 Guangzhou Medical Expo Scheduled for August! Building the Core Engine for the Greater Bay Area's Healthcare Industry. 2025-11-07
- Full-chain Empowers Industrial Innovation! The 2025 Guangzhou Medical Expo Successfully Concludes Today 2025-08-24
- Grand Opening of 2025 Guangzhou Medical and Health Industry Expo: Jointly Paint a New Blueprint for Health and Play the March of Industrial Endeavor 2025-08-22
- Over 400 hospitals and enterprises gather in the City of Rams! Guangzhou Medical Expo has become a highland for the first launches of innovative pharmaceuticals and medical devices. 2025-06-13